CoNextions TR Zone 4 Glide White Paper
Objective
The purpose of this study was to compare the cross-sectional area of the tendon repair site and glide forces obtained during simulated tendon motion through the carpal tunnel before and after repairs made with the CoNextions TR Tendon Repair System or a conventional suture repair technique.
Background
Suture has remained the standard of care for the repair of lacerated or severed tendons since it was first documented over 1800 years ago (Manske 2005). Despite advancements in biomaterials, operative techniques, and rehabilitation protocols, postoperative complications following tendon repair are still common. One of the prevailing principles of tendon repair is to reduce the additional bulk at the repair site as bulkier repairs have been shown to impede normal tendon gliding (Zhao 2004).
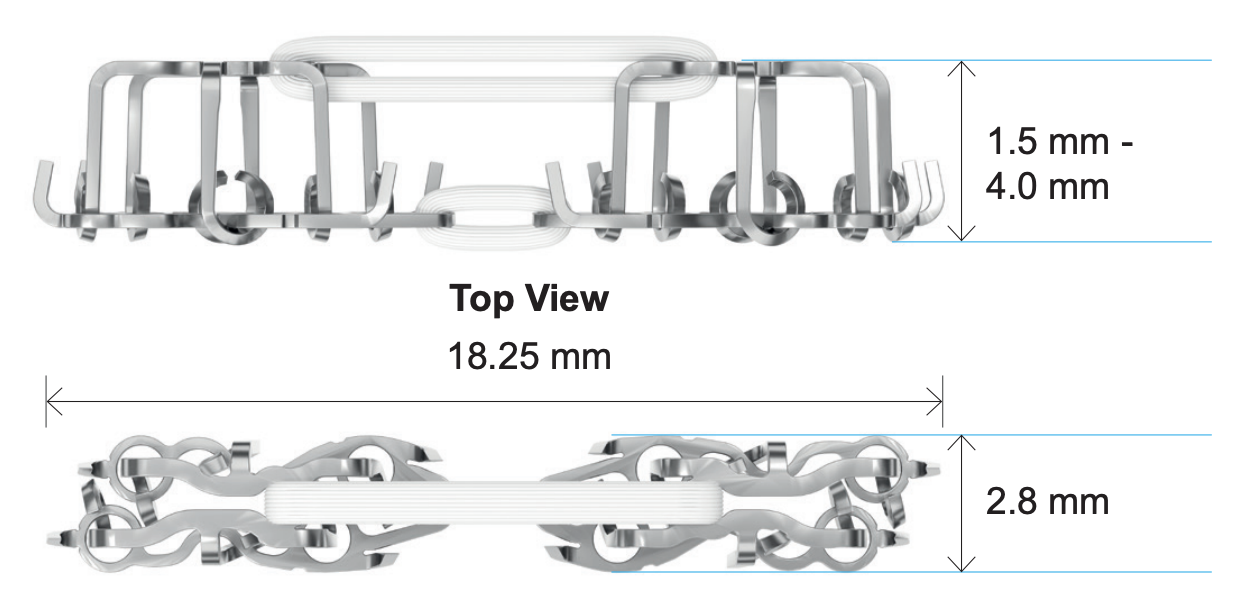
The CoNextions TR Tendon Repair System was designed to provide an alternative to suture for the repair of tendons. The implant consists of two identical stainless steel anchors implanted simultaneously into the injured tendon. The tendon repair site is centered between the anchors which are connected by two loops of ultra-high molecular weight polyethylene (UHMWPE) fiber (Figure 1).
The CoNextions TR implant is provided in two pieces pre-loaded into an implant mechanism. During implant deployment the top and bottom pieces of the implant form together securing the reapproximated ends of the injured tendon. The system was designed for the repair of tendons that are 1.5 to 4 mm in thickness and at least 3 mm in width, and requires at least 20 mm (10 mm/side) of surgical site access for proper implant deployment.
Specimen Preparation
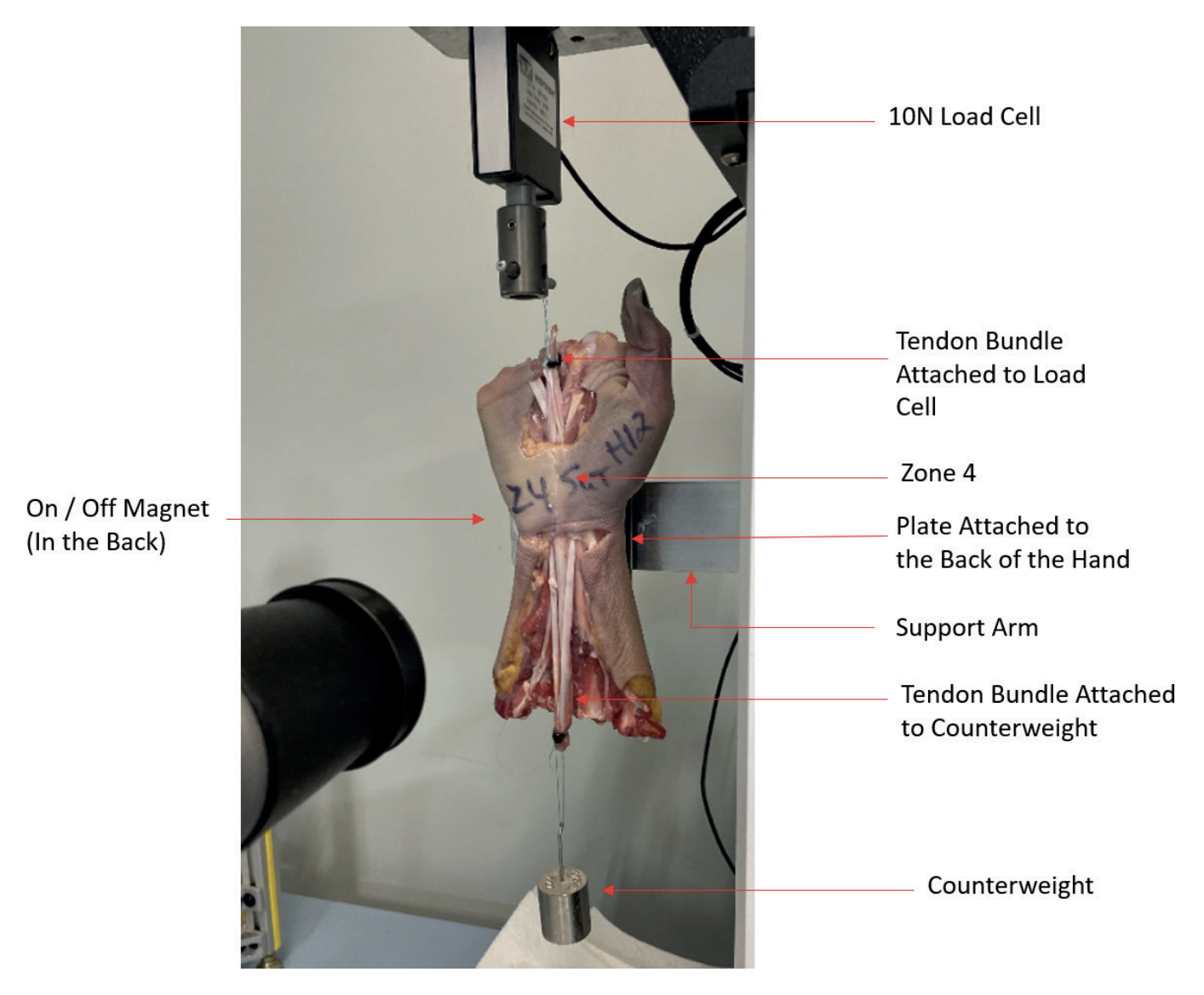
Cadaveric hands were dissected to free flexor tendons superficialis (FDS) of the index, long and ring digits from surrounding tissue proximal and distal to the carpal tunnel (Zone 4). The adjacent flexor digitorum profundus (FDP) tendons and median nerves were trimmed close enough to either side of the carpal tunnel to allow them to remain in the tunnel during the simulated tendon motion. The digits of the specimens were removed at the metacarpophalangeal (MCP) joint to prevent the fingers from interfering with the tensile test. A metal plate was attached to the back of the specimen at the phalangeal, ulna, and radius bones to fix the specimen to the support arm for tensile testing. The tendons of the index, ring, and long digit to create a tendon bundle. Sutures were placed proximally and distally to create loops that could be used to connect one side of the tendon bundle to the load cell of the test frame and the other side of the tendon bundle to a 100 g counterweight (Figure 2).
Repair Measurements
The width and height of each FDS tendon was measured with digital calipers prior to and after the simulated repairs. These measurements were used to calculate an estimated cross-sectional area (CSA) of the repair, defined as the approximate area of an ellipse.
Glide Testing
Each specimen was attached to the support arm using the plate and magnet described above for native glide force testing. The specimens were oriented so the line of pull of the tendon bundle could be pulled in tension, and the tendon bundle was as free to glide as possible.
Initially the specimen was placed with the hand up as shown in Figure 2. Tendon bundles were connected to the load cell (10N) and to the counterweight and an Instron® tensile testing machine. (Instron 3342 Series Universal Testing Device, Instron Corporation) was used to apply load to the bundles and pull them distally through the carpal tunnel.
The specimen was then rotated 180° and the test was repeated to pull the tendon bundles proximally through the carpal tunnel. After completing the native glide test, one of the FDS tendons in the bundle was repaired with either a CoNextions TR Implant or a 4-strand locked cruciate suture repair (Ethibond Excel® 3-0 Braided Polyester Suture) by a fellowship-trained hand surgeon and the tensile Figure 2: Zone 2 Glide Force Test Setup tests were repeated. This entire process was repeated until all three FDS tendons in the tendon bundle had been repaired and tensile tested during simulated tendon motion.
Results
Cross-Sectional Area of Tendon Repairs
The average percent increase in the CSA of tendons repaired with a CoNextions TR implant was 42% compared to an average increase of 105% in the CSA of tendons repaired with suture (Table 1).
Glide Force Testing
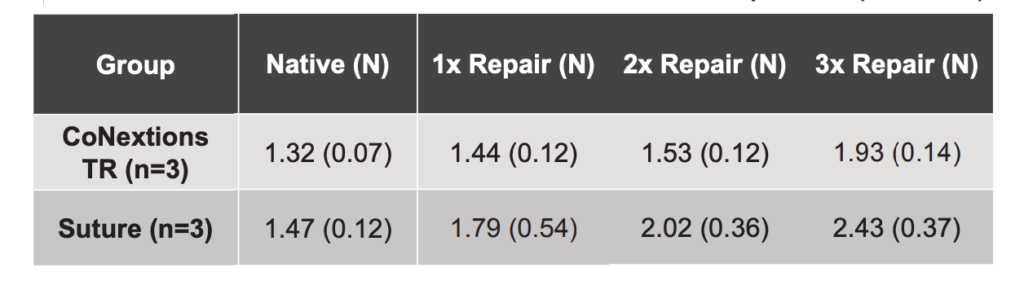
FDS tendon bundles repaired with a CoNextions TR implant had lower maximum glide forces than tendons repaired with suture during simulated glide through the carpal tunnel when 1x, 2x, and 3x of the FDS tendons had been repaired (Table 2).

and 3x CoNextions TR and Suture Repairs
Conclusion
FDS tendons repaired with a CoNextions TR implant had a smaller increase in their cross-sectional area than matched FDS tendons repaired with a 4-strand locked cruciate suture pattern. The CoNextions TR repairs also saw lower glide forces than suture repairs during simulated tendon gliding through the carpal tunnel.
References
- Manske P. History of flexor tendon repair. Hand Clin 2005;221(2):123-7.
- Zhao C, et al. Effect of gap size on gliding resistance after flexor tendon repair. JBJS (Am) 2004;86(11):2482-8.
Other Reads:
CoNextions TR Zone 2 Glide White Paper | Clinical Trail Summary | Biomechanical Comparison of Repair Strength
This document is intended solely for the use of healthcare professionals. Benchtop testing may not be indicative of clinical performance. The information presented is intended to demonstrate a CoNextions Inc. product. CoNextions does not practice medicine and recommends that surgeons be trained in the use of any particular product before using it in surgery. A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. CoNextions does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery. Individual results will vary based on health, weight, activity and other variables. Not all patients are candidates for this product and/or procedure. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your CoNextions representative if you have questions about the availability of CoNextions products in your area. For indications, contraindications, warnings, precautions, potential adverse effects and patient counselling information, see the package insert or contact your local representative. Visit www.conextionsmed.com for additional product information. CoNextions owns, uses, or has applied for the following trademarks: CoNextions, Coronet, Revolutionizing Tendon Repair. Sawbones is a trademark of Pacific Laboratories Inc. FiberWire is a trademark of Arthrex, Inc. Instron is a trademark of Illinois Tool Works Inc.
MC0010.A
© 2021 CoNextions Inc. All Rights Reserved. U.S and Foreign Patents Pending. Patented in the U.S. Patent No. 10,219,804.
