CoNextions ASSH Abstract White Paper
The following abstract won the Joseph H. Boys Award at the 39th Annual Adrian E. Flatt Residents and Fellows Conference during the 76th Annual Meeting of the American Society for Surgery for the Hand in San Francisco, California (September 30-October 2, 2021).
Tendon Repairs at the Wrist Utilizing a Novel Tendon Stapler Device: An Efficiency and Biomechanical Study Across Different Experience Levels
Evelyn R Reed, MD, Russell Hendrycks, BS, Emily M. Graham, BSN, Megan Rosales, MS, MStat, and Shaun D. Mendenhall, Plastic and Reconstructive Surgery, University of Utah, Salt Lake City, Utah.
HYPOTHESIS
We hypothesized a novel tendon stapler device (TSD), the CoNextions Tendon Repair System, would exhibit decreased repair time and superior biomechanical force profiles when compared to standard core suture for primary tendon repairs.
METHODS
Participants included a novice, intermediate, and expert at tendon repairs. Comparisons were performed on 6 left and 6 right matched human cadaver arm (12 total) finger and wrist extensors and flexors. Novice and intermediate participants received suture repair training from the expert, and all participants received equal TSD training. Suture repairs were performed with a 3.0 braided polyester modified Kessler with a horizontal mattress. TSD repairs were done on matched donor arms with a single simple polypropylene 5.0 stitch to approximate the tendon ends prior to device deployment. Repairs were timed by a nonparticipating data recorder. Immediately after repair, biomechanical testing was performed to compare tensile strength (2mm gap force) and ultimate failure loads with mode of failure noted. Statistical analyses utilized one-way ANOVA, Tukey pairwise comparisons, two-sided independent samples ttest, and Fisher’s exact test.
RESULTS
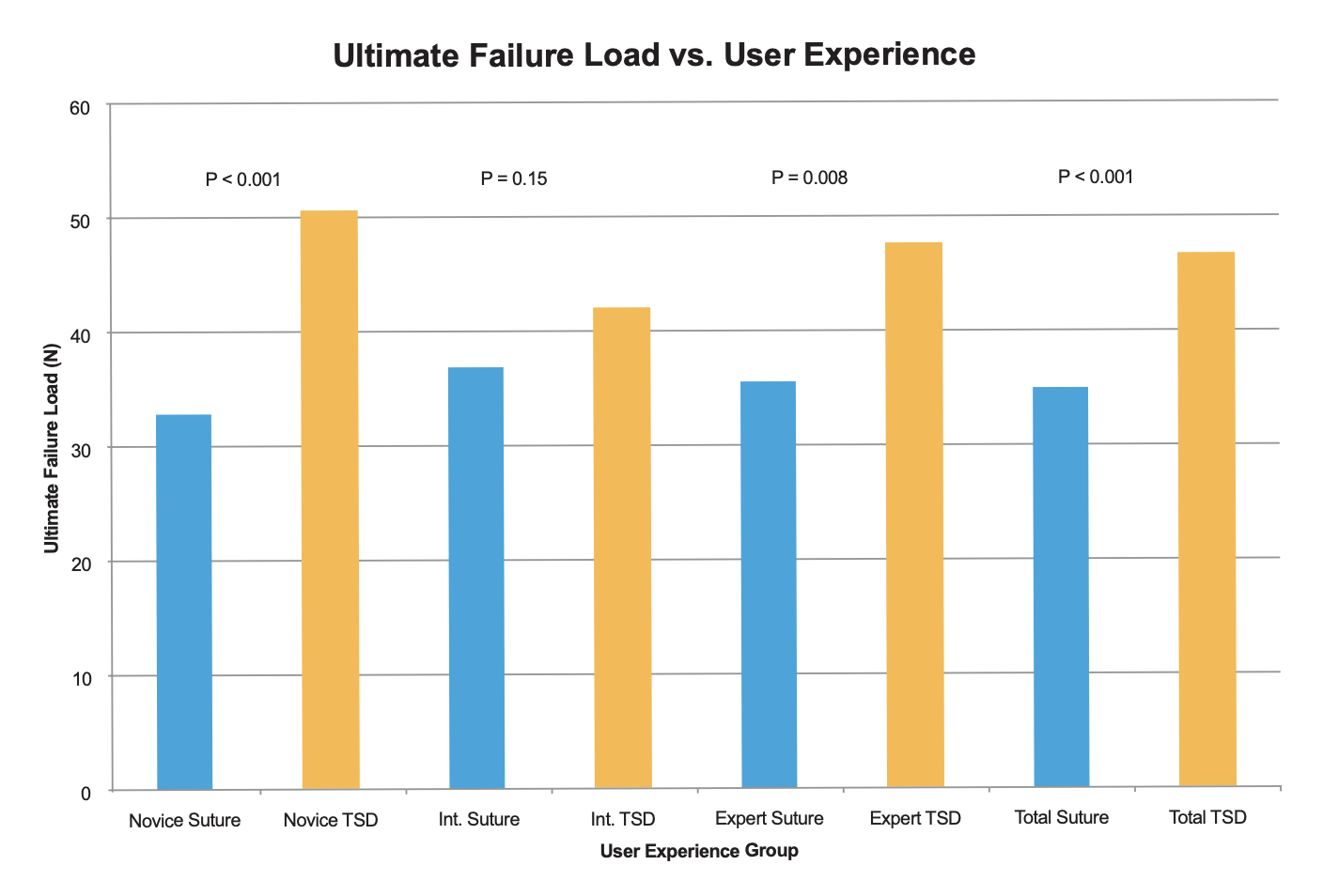
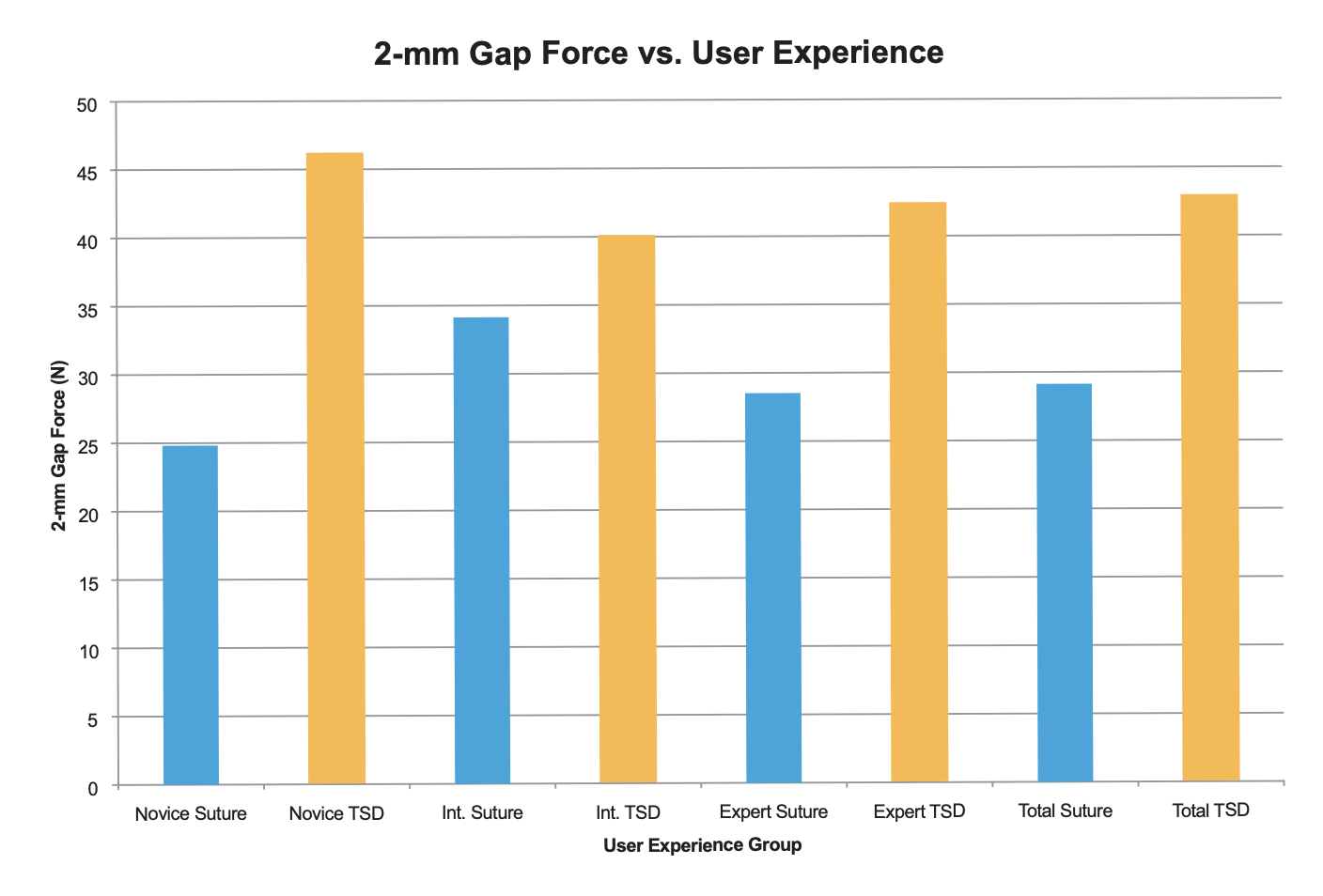
In total, 228 tendon repairs were analyzed. Suture tendon repair times were significantly lower for expert (3.25 minutes) than intermediate (4.08 minutes) and novice (4.46 minutes) (p<0.01). TSD mean repair times were 1.32, 1.31, and 1.28 minutes for expert, intermediate, and novice groups respectively with no difference between groups. Tendon repairs performed with the TSD were significantly faster (p<0.01) across all experience groups. Tensile strength to a 2mm gap and ultimate failure load were statistically higher in the TSD group for the expert and novice participants (p<0.01), and directionally higher for the intermediate (p=0.09). There was no difference in gap and ultimate tensile strength between participants using the TSD device. Compared to the TSD, the suture method had significantly higher proportions of material failures (knot or suture failure) and a lower proportion of pull-through failure (p<0.01).
- Compared to standard suture repair, the TSD repair is more efficient and produces strongertendon repairs among users of varying experience.
- This device has the potential to be extremely helpful in scenarios where multiple tendonrepairs are required, such as spaghetti wrists and hand replantation or transplantation.
- Forthcoming studies will explore how the TSD performs in in-vivo.
Other Reads:
Clinical Trial Summary | CoNextions TR Zone 2 White Paper | Biomechanical Comparison of Repair Strength
This document is intended solely for the use of healthcare professionals. Benchtop testing may not be indicative of clinical performance. The information presented is intended to demonstrate a CoNextions Inc. product. CoNextions does not practice medicine and recommends that surgeons be trained in the use of any particular product before using it in surgery. A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. CoNextions does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery. Individual results will vary based on health, weight, activity and other variables. Not all patients are candidates for this product and/or procedure. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your CoNextions representative if you have questions about the availability of CoNextions products in your area. For indications, contraindications, warnings, precautions, potential adverse effects and patient counselling information, see the package insert or contact your local representative. Visit www.conextionsmed.com for additional product information. CoNextions owns, uses, or has applied for the following trademarks: CoNextions, Coronet, Revolutionizing Tendon Repair. Sawbones is a trademark of Pacific Laboratories Inc. FiberWire is a trademark of Arthrex, Inc. Instron is a trademark of Illinois Tool Works Inc.
MC0010.A
© 2021 CoNextions Inc. All Rights Reserved. U.S and Foreign Patents Pending. Patented in the U.S. Patent No. 10,219,804.
