Clinical Trial Summary White Paper
Michael Solomons1, Antoine Rocher2, Ajmal Ikram3, Grant Biddulph4, and Duncan McGuire1
- Groote Schuur Hospital, Cape Town, South Africa
- Inkosi Albert Luthuli Central Hospital, Durban, South Africa
- Chris Hani Baragwanath Academic Hospital, Soweto, South Africa
- Tygerberg Hospital, Cape Town, South Africa
Introduction
Suture has remained the standard of care for the repair of lacerated or severed tendons since it was first documented over 1800 years ago (Manske 2005). Despite advancements in biomaterials, operative techniques, and rehabilitation protocols, postoperative complications following tendon repair are still common. These complications include rupture of the tendon repair and adhesion formation at the repair site and can lead to long-term restriction of the joint mobility, additional surgical procedures, and extensive rehabilitation (Dy 2012). Injuries of the flexor digitorum profundus (FDP) tendons in Zone 2 are particularly challenging due to the unique anatomy of this location (Bunnell 1918, Griffin 2012).
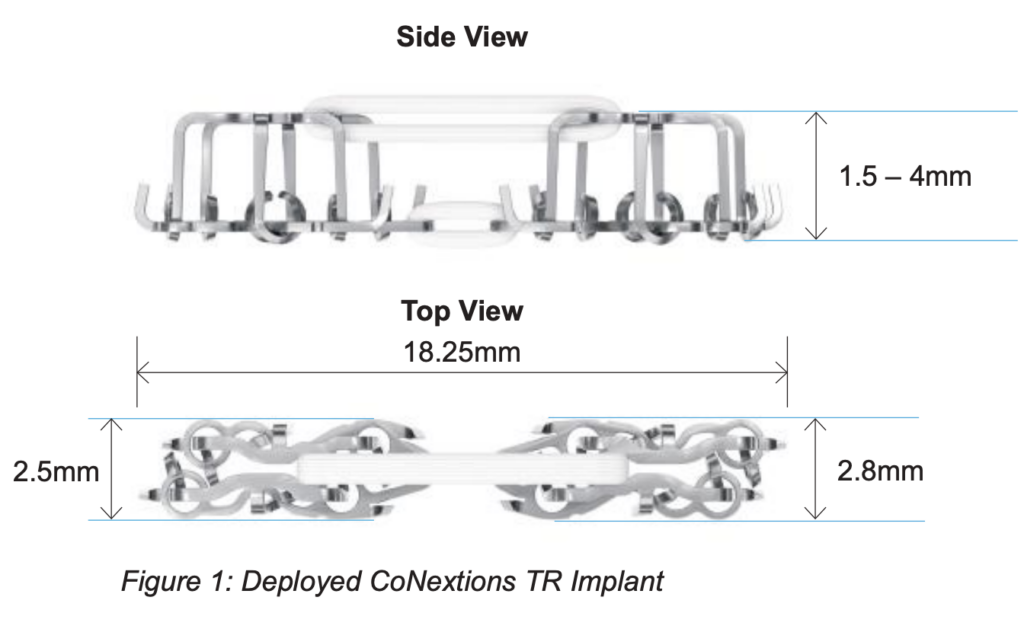
The CoNextions TR® Tendon Repair System (“CoNextions TR”) was designed to provide an alternative to suture for the repair of tendons. The implant consists of two identical stainless steel anchors implanted simultaneously into the injured tendon. The tendon repair site is centered between the anchors which are connected by two loops of ultra-high molecular weight polyethylene (UHMWPE) fiber (Figure 1).
The CoNextions TR implant is provided in two pieces preloaded into an Implant Mechanism. During implant deployment the top and bottom pieces of the implant form together securing the reapproximated ends of the injured tendon. The system was designed for the repair of tendons that are less than 4 mm in thickness. The system requires at least 20 mm (10 mm/side) of surgical site access for proper implant deployment.
In various bench testing and animal studies, CoNextions TR performed at least as well suture for a number over performance metrics.
The following clinical study was performed at the behest of regulatory bodies in the United States and Europe and was designed to provide a comparative assessment of the safety and eƯectiveness of CoNextions TR and a suture repair technique for the repair of lacerated Zone 2 FDP tendons.
Study Design
The study was performed in compliance with International Conference on Harmonization Good Clinical Practices, received approval from South African Health Products Regulatory Authority and the individual institutional review boards, and was monitored by an independent data and safety board.
Over the course of two and a half years, 5 orthopaedic hand surgeons located at 4 sites in South Africa enrolled 90 participants presenting with lacerations of one or two flexor digitorum profundus (FDP) tendons in Zone 2. Following consent and eligibility determination, the participants were randomly assigned intraoperatively to have their injury repaired using CoNextions TR or a four strand locked cruciate repair (3.0 or 4.0 PROLENE® Polypropylene Suture, Ethicon, Inc.) with a running epitenon suture (6.0 nylon suture).
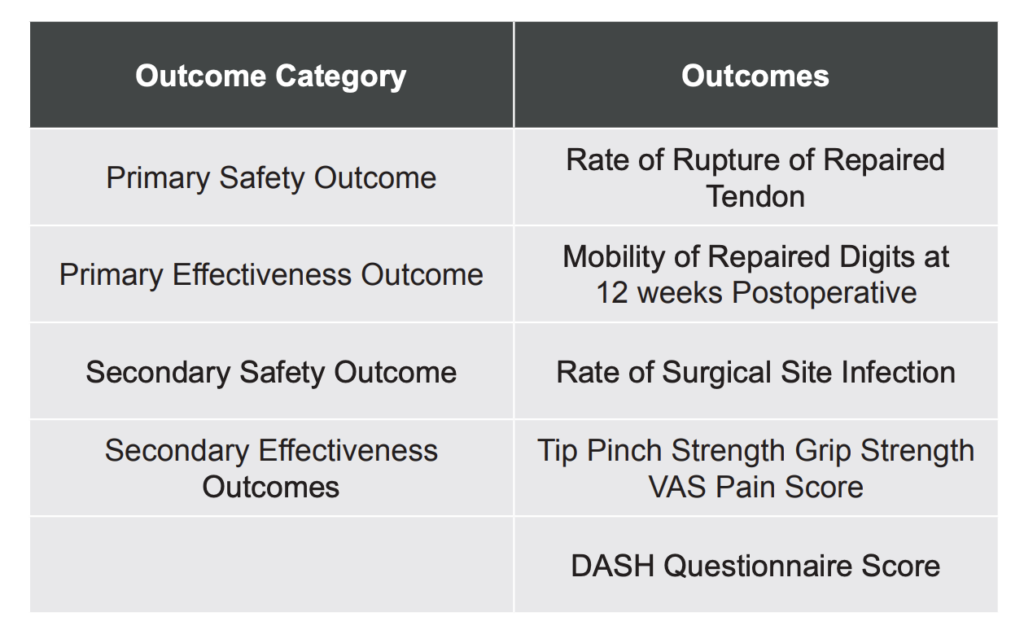
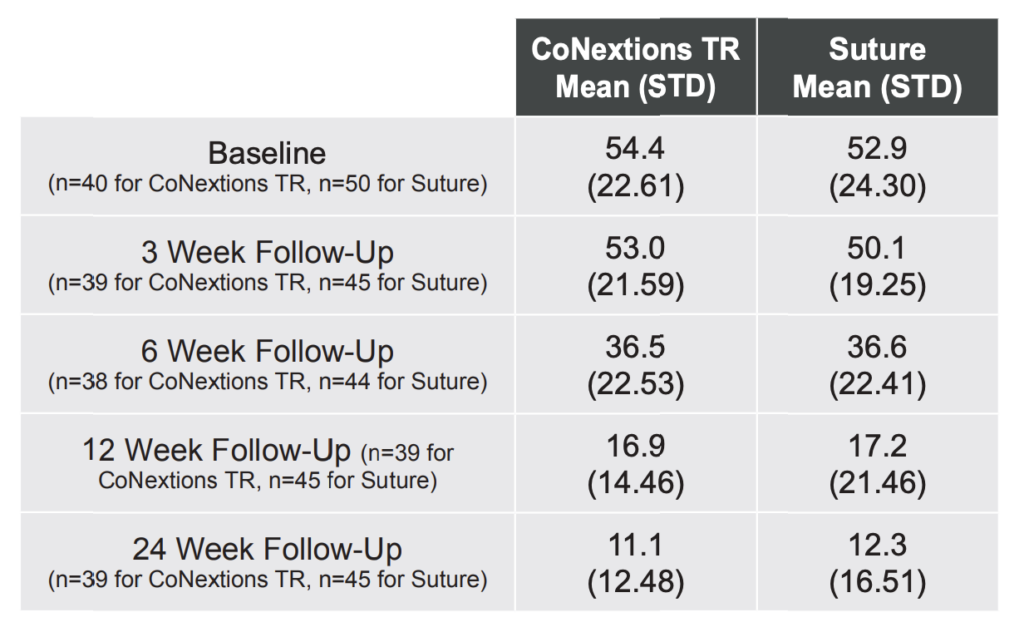
Participants were followed for 24 weeks postoperative with the primary safety endpoint for the study being the rate of tendon rupture following the surgical repair and the primary effectiveness endpoint of the study being the mobility of the repaired digits at 12 weeks postoperative (using Strickland Mobility Scores) (Strickland 1985). The rate of surgical site infection (SSI) served as a secondary safety outcome. Tip pinch strength and grip strength measurements were collected as secondary measures of postoperative digital function. Data related to the participant’s perception of their pain (VAS Pain Score, 0-10 cm scale) and disability (Disability of the Arm, Shoulder and Hands, DASH Questionnaire) were also collected as secondary outcome measures (Table 1).
A four-strand locked cruciate (3.0 or 4.0 PROLENE® Polypropylene Suture, Ethicon, Inc.) and a running epitenon suture (6.0 nylon suture) was used for the tendon repairs for participants randomized to the Suture group. All participants were prescribed a standardized postoperative rehabilitation protocol for the first 12 weeks postoperative. An occupational therapist overseeing the rehabilitation protocol collected the majority of the outcome assessments, with both the therapist and participant blinded to the assigned treatment arm for the first 12 weeks postoperative
Results
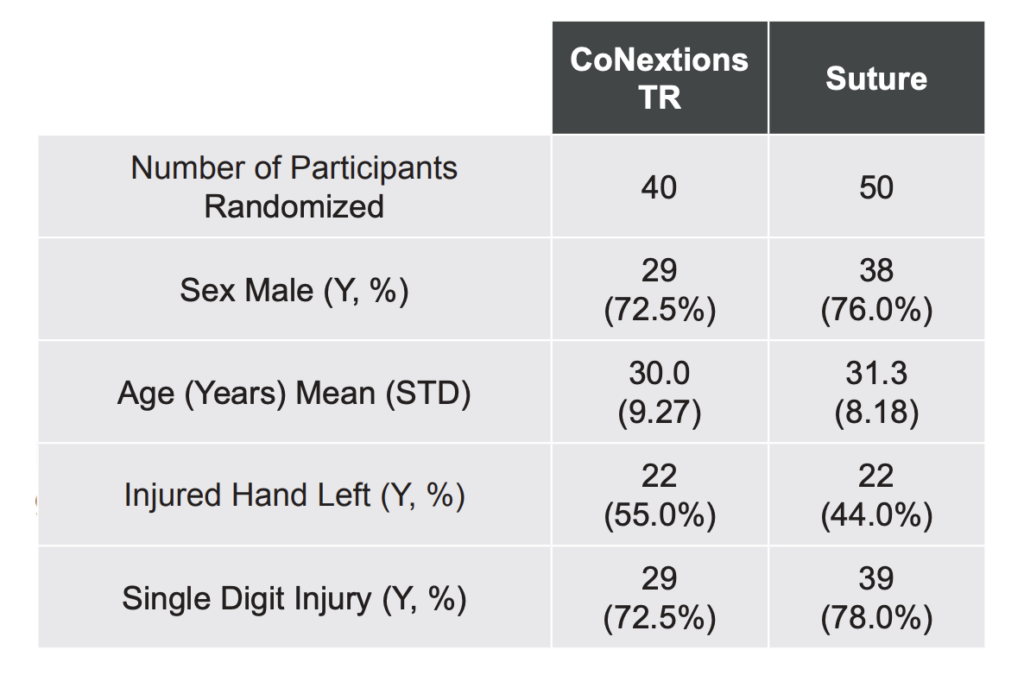
Participant Demographics: 40 participants were randomized to the CoNextions TR group and 50 participants were randomized to the Suture group (Table 2).
None of the demographic differences between the two groups was statistically significant.
Rate of Re-Rupture of Repaired Digit(s): One (1, 2.56%) of 39 participants in the CoNextions TR group presented with a rupture of the repaired digit with one rupture occurring in 51 repaired digits out (1.96%). Five (5, 10.42%) of 48 participants in the Suture group presented with a rupture of the repaired digit(s) with 6 ruptures occurring in the 61 repaired tendons (9.84%) (Figure 2).
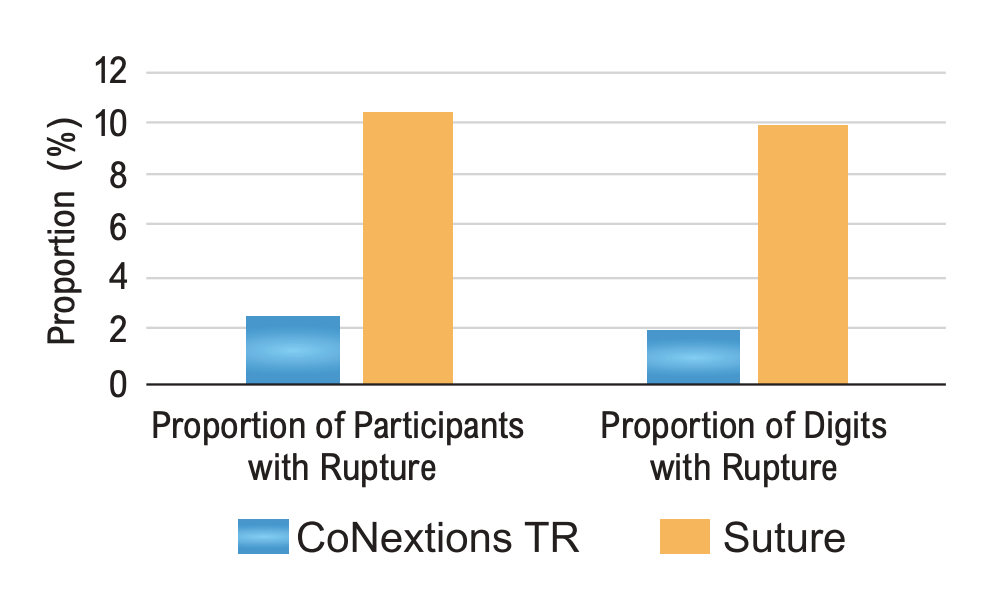
Although statistical superiority was not shown for this endpoint, the lower rupture rate in the
CoNextions TR group is notable. Given rupture represents a definitive failure of the original repair, the difference in rates observed between the two groups in this study is likely of clinical importance.
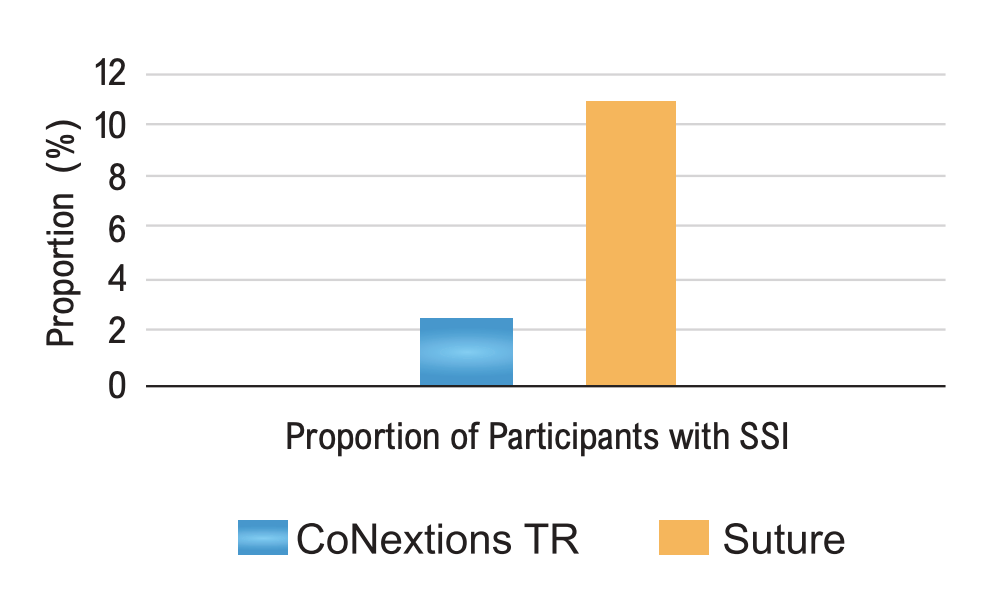
Rate of Surgical Site Infection: One (1, 2.70%) of 37 participants in the CoNextions TR group experienced an SSI with 5 of 45 (11.11%) participants in the Suture group experiencing an SSI (Figure 3).
Although this difference did not represent a
statistically significant difference, the difference in rates observed between the two groups is likely of clinical importance.
Mobility of Repaired Digit(s): Strickland mobility scores were calculated using the following formula with 175 representing a normal value for an uninjured finger:
Strickland Score = 100 * (total active flexion – total extension deficit (PIP, DIP)/175)
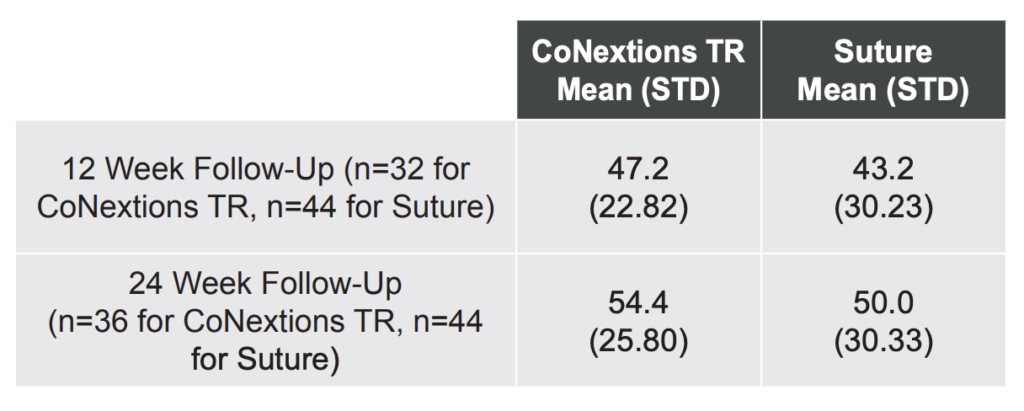
Strickland mobility scores were comparable for the two groups at the 12, and 24 week follow-up visits with the average score in both groups improving over time (Table 3, Figure 4).
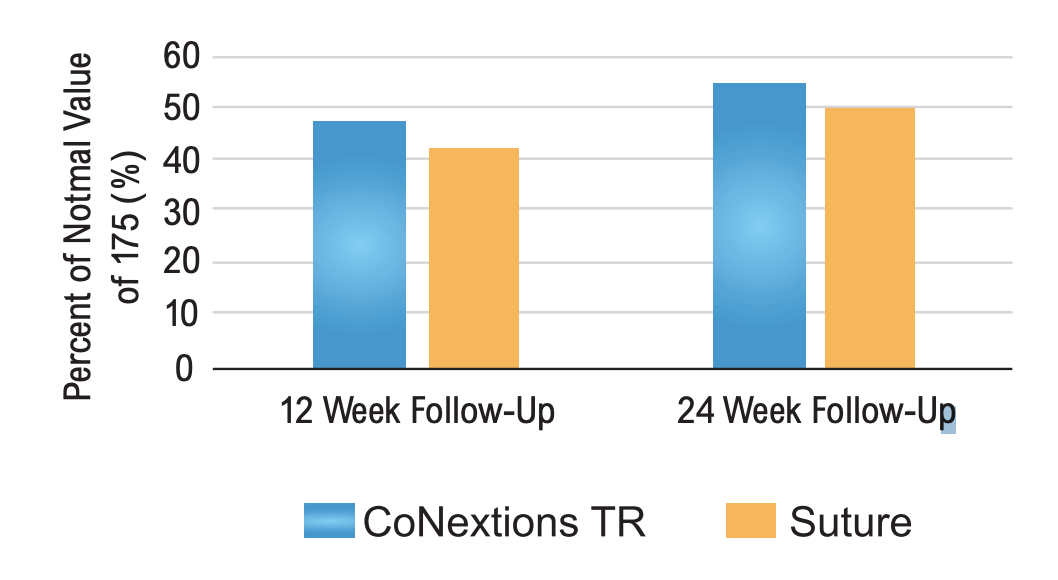
Note: Data presented is for the research index digit with no imputation for lost values. If 12 week data are not present due to a rupture, a score of Zero is imputed
None of the differences between the two groups were statistically significant. Any differences
between the two groups were less than the minimal detectable difference for finger goniometry
(Reissner 2019).
Tip Pinch Strength: Tip pinch strengths were comparable between the two groups at the 12 and 24 week follow-up visits with the average score in both groups improving over time (Table 4, Figure 5).
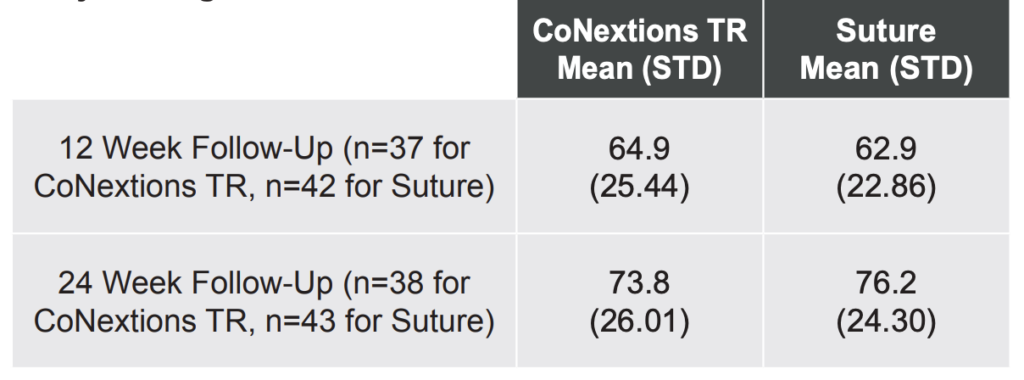
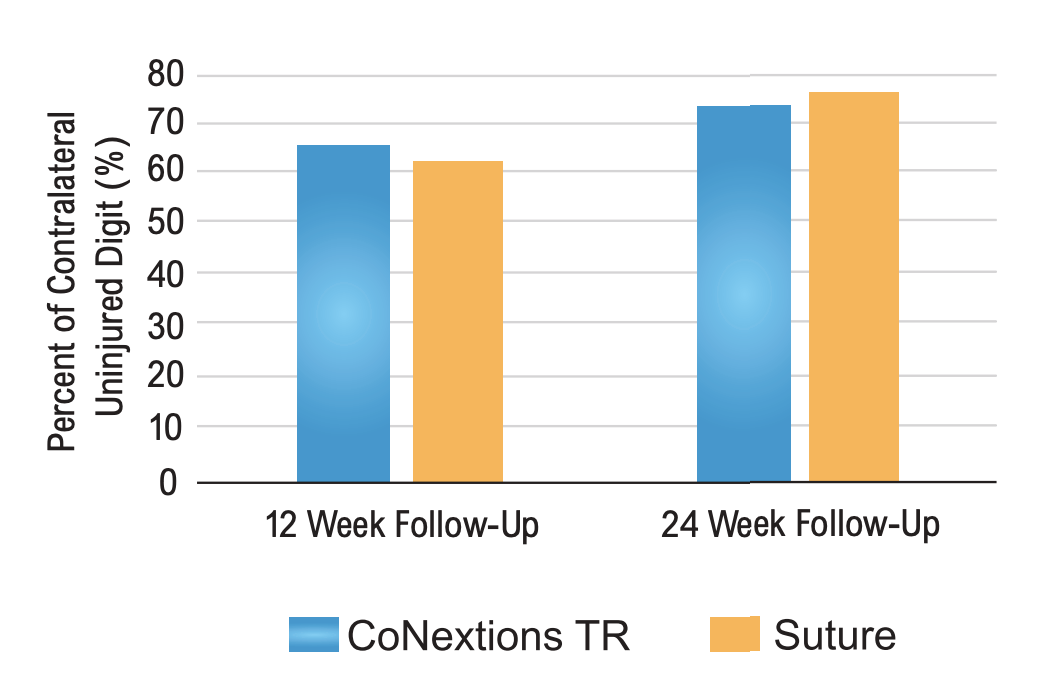
None of the differences between the two groups were statistically significant. Any differences between the two groups were less than the minimal clinically important difference (MCID) of 7.7% determined by Villafane, et al. (2017).
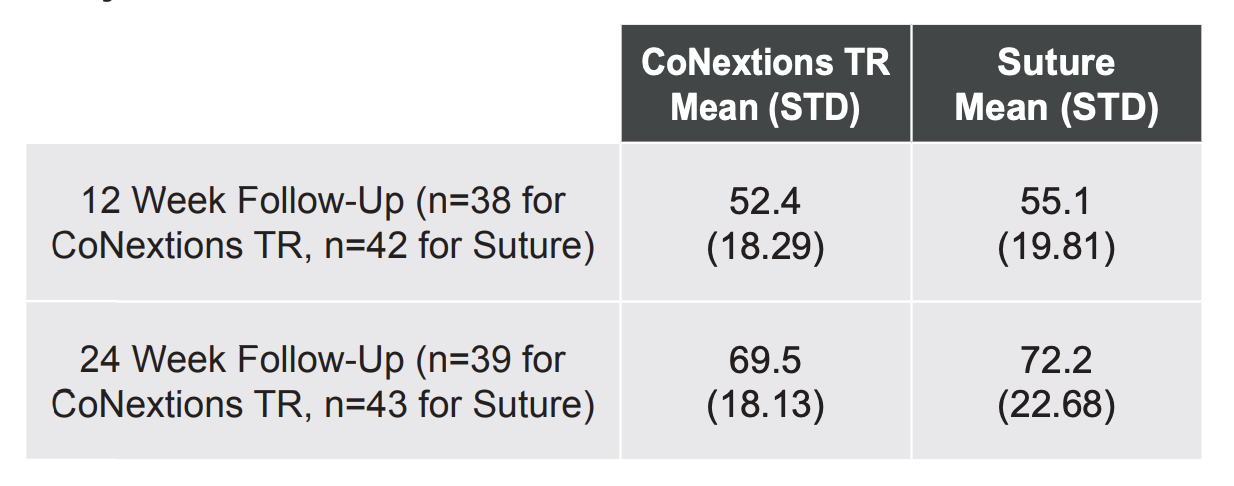
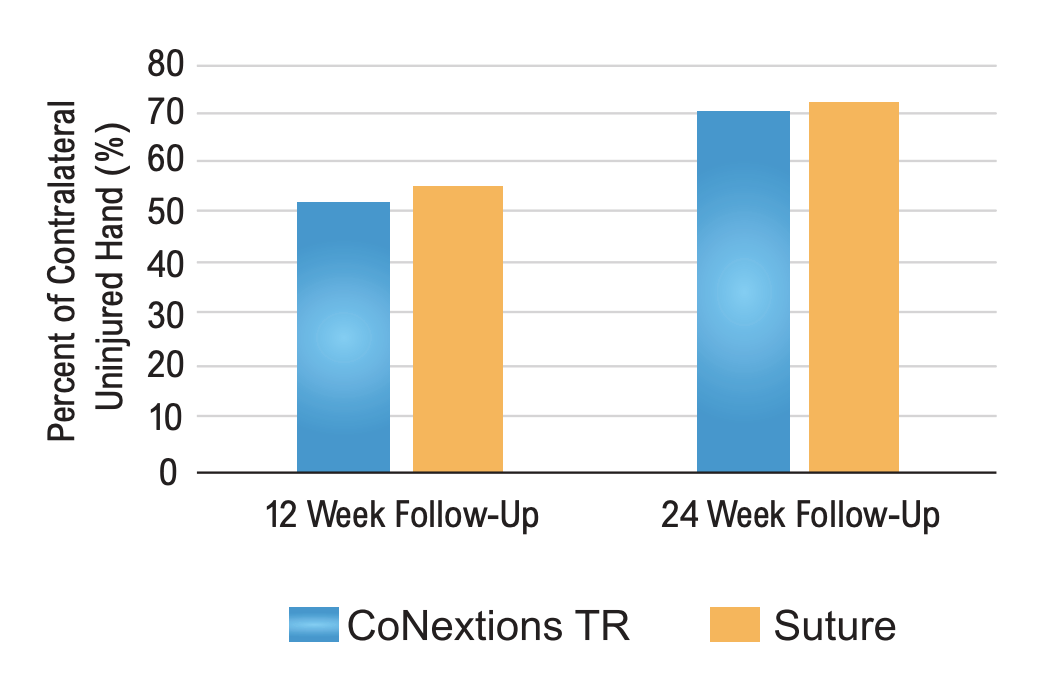
Grip Strength: Grip strengths were comparable between the two groups at the 12 and 24 week followup visits with the average score in both groups improving over time (Table 5, Figure 6).
Uninjured Hand
Uninjured Hand
None of the differences between the two groups were statistically significant. Any differences
between the two groups were less than the reported MCID of 19.5% (Kim 2014).
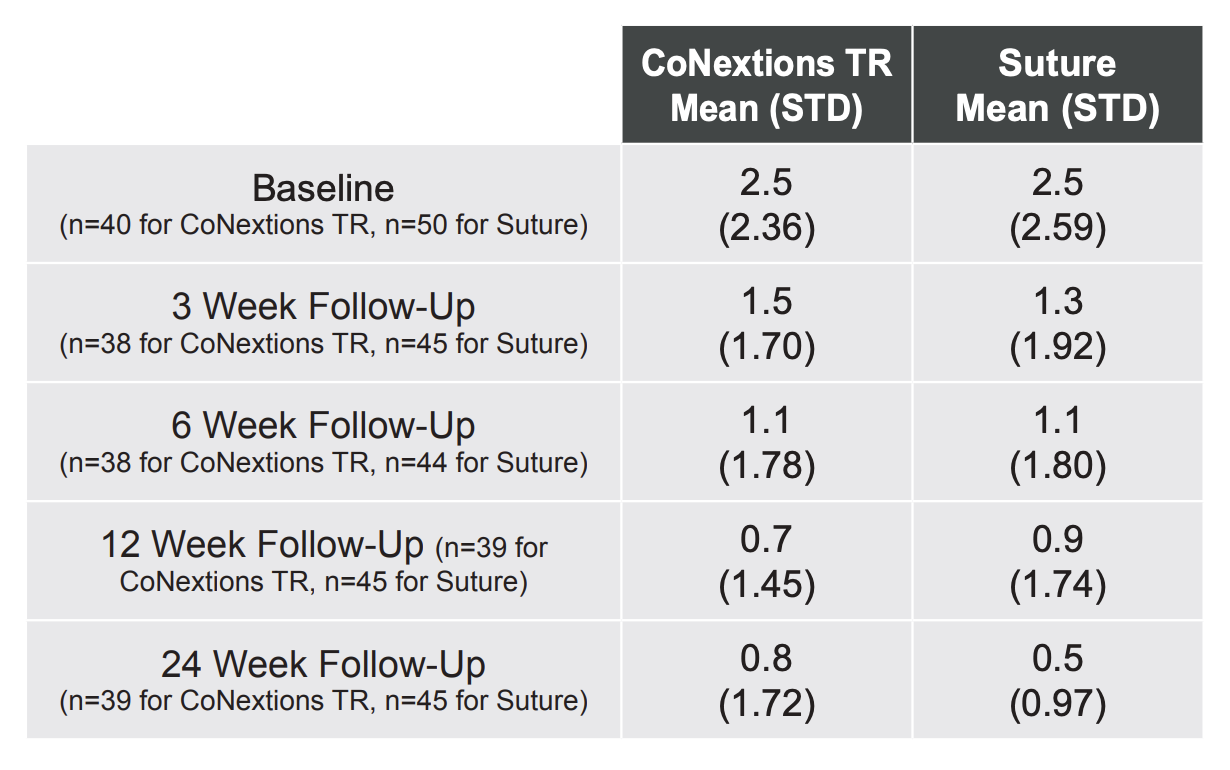
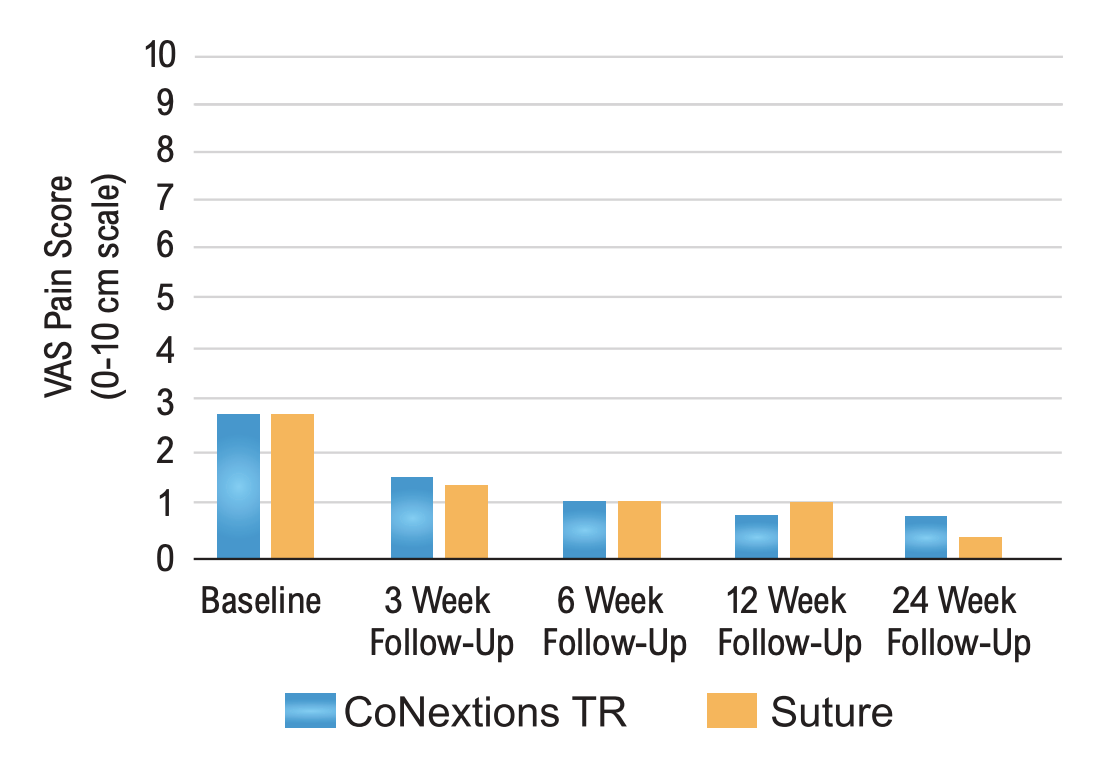
VAS Pain Scores: VAS Pain scores were comparable between the two groups at all time points with the average score in both groups decreasing over time (Table 6, Figure 7).
Uninjured Hand
None of the differences between the two groups were statistically significant. Any differences between the two groups for a given time point were smaller than the MCID of 1.4 cm determined by Tashjian et al (2009).
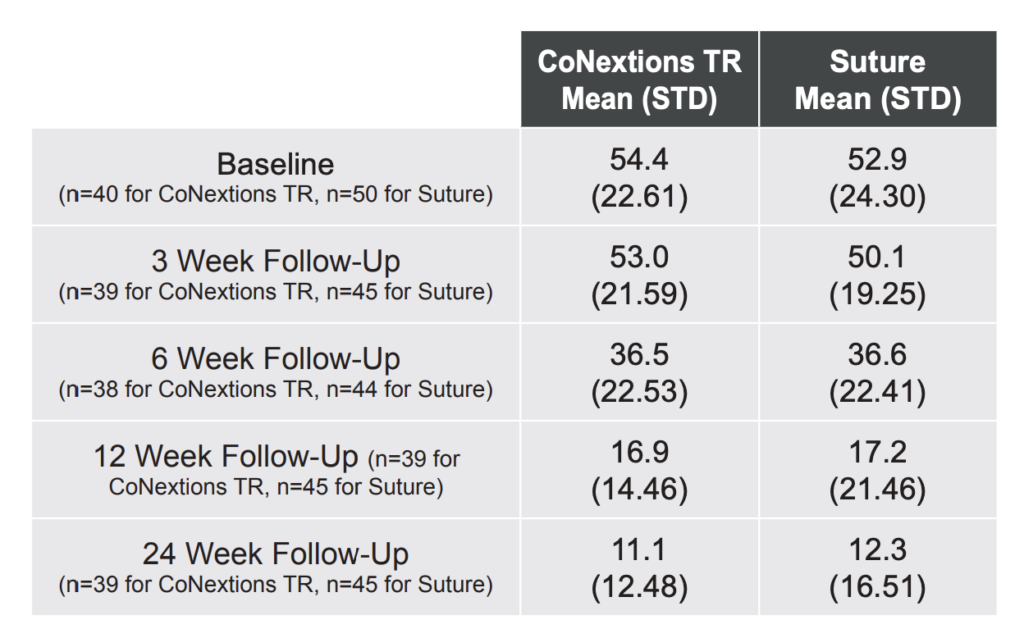
DASH Questionnaire Scores: DASH Questionnaire scores (utilizing a 0 (no disability) to 100 (complete disability) scale) were comparable between the two groups at all time points with the average scores in both groups improving (decreasing) over time (Table 7, Figure 8).
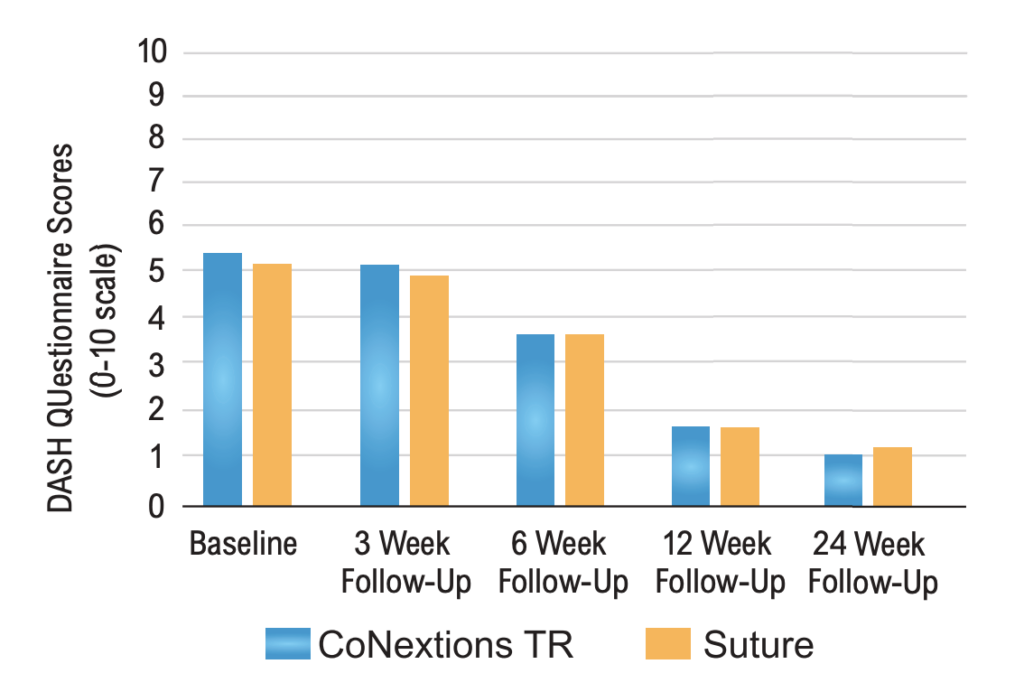
None of the differences between the two groups were statistically significant. Any differences between the two groups for a given time point were smaller than the reported MCID of 10.81 (Franchignoni 2013).
Safety Monitoring: There were no participant deaths during the conduct of the study. The safety reporting was comparable for the two groups with the CoNextions TR group having a lower rate of adverse (AE) and serious adverse (SAE) events per participant (Table 8).
For both groups, the most commonly reported AEs related to the injured digit(s) were postoperative pain, swelling, stiƯness and neuralgia. These AEs are understood to be common following orthopaedic surgery and are frequently interrelated in their presentation (Gagnier 2017).
Conclusion
For this study, a lower rate of rupture of the repaired tendons and a lower rate of SSI was observed in the CoNextions TR group compared to the Suture group. Though neither difference was statistically significant, the observed differences were substantial enough to likely be of clinical importance. The overall safety monitoring demonstrated a lower rate of AE and SAE in the CoNextions TR group compared to the Suture group. None of known risks of tendon repair were seen to occur with unexpected frequency in either group.
For the primary effectiveness endpoint of mobility of the repaired digits, the two groups were comparable with any differences between them being less than the minimum detectable difference of finger goniometry. Tip Pinch Strength, Grip Strength, VAS Pain Scores and DASH Questionnaire Scores were comparable between the two groups at all time points with any differences between the two groups being smaller than the respective MCID for the outcome measure. The lack of significant differences between the two groups for the various effectiveness outcomes was an expected outcome for this study given a standardized rehabilitation program was used. This finding is also consistent with the literature, where the use of different primary suture techniques has not been demonstrated to result in statistically significant or clinically meaningful differences in effectiveness outcomes.
The results of this clinical study indicate CoNextions TR is at least as safe and effective as suture for the repair of lacerated Zone 2 FDP tendons.
References
Bunnel S. Repair of tendons in the fingers and description of two new instruments. Surg Gynecol Obstet 1918;26:103-10.
Dy CJ, et a. Complications after flexor tendon repair: a systematic review and meta-analysis. J Hand Surg AM 2012;37A:543-51.
Franchignoni F, et al. Minimal clinically important difference of the disabilities of the arm, shoulder and hand outcome measure (DASH) and its shortened version (QuickDASH). J Orthop Sports Phys Ther 2013;44(1):30-39.
Gagnier JJ et al. A retrospective cohort study of adverse events in patients undergoing orthopaedic surgery. Patient Saf Surg 2017;11(15).
Griffin M, et al. An overview of the management of flexor tendon injuries. Open Orthopaedics Journal 2012;6:28-35.
Kim JK, et al. What is the minimum clinically important difference in grip strength. Clin Orthop Relat Res 2014;472(8):2536-41.
Manske PR. History of Flexor Tendon Repair. Hand Clin 2005;21:123-7.
Reissner L, et al. Minimal detectable difference of the finger and wrist range of motion: comparison of goniometry and 3D motion analysis.
Journal of Orthopaedic Surgery and Research 2019;173.
Strickland JW. Opinions and preferences in flexor tendon surgery.
Hand Clin 1985;1:167-79.
Tashjian RZ, et al. Minimal clinically important differences (MCID) and patient acceptable symptomatic state (PASS) for visual analog scales (VAS) measuring pain in patients treated for rotator cuff disease.
J Shoulder Elbow Surg 2009;18:927-32.
Villafane JH, et al. Minimal clinically important difference of grip and pinch strength in women with thumb carpometacarpal osteoarthritis when compared to healthy subjects.
Rehabil Nurs 2017;42(3):139-45.
More information regarding this study is publicly available at http://clinicaltrials.gov/ct2/show/nct03622372
Other Reads:
This document is intended solely for the use of healthcare professionals. Benchtop testing may not be indicative of clinical performance. The information presented is intended to demonstrate a CoNextions Inc. product. CoNextions does not practice medicine and recommends that surgeons be trained in the use of any particular product before using it in surgery. A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. CoNextions does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery. Individual results will vary based on health, weight, activity and other variables. Not all patients are candidates for this product and/or procedure. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your CoNextions representative if you have questions about the availability of CoNextions products in your area. For indications, contraindications, warnings, precautions, potential adverse effects and patient counselling information, see the package insert or contact your local representative. Visit www.conextionsmed.com for additional product information. CoNextions owns, uses, or has applied for the following trademarks: CoNextions, Coronet, Revolutionizing Tendon Repair. Sawbones is a trademark of Pacific Laboratories Inc. FiberWire is a trademark of Arthrex, Inc. Instron is a trademark of Illinois Tool Works Inc.
MC0010.A
© 2021 CoNextions Inc. All Rights Reserved. U.S and Foreign Patents Pending. Patented in the U.S. Patent No. 10,219,804.
